Teacher: Annika Singh
SEMESTER 6
C13 RECOMBINANT DNA TECHNOLOGY
Paper Code (BBT 6001)
(Credits: Theory-4, Practicals-2)
THEORY Lectures: 40
Course Objective(s)
· Recall different DNA modifying enzymes used in recombinant DNA technology
· Compare different vectors and their applications in recombinant DNA technology
· Illustrate different techniques used in genetic engineering
Course Outcome(s):
· Students will take practical training in the recent techniques of recombinant DNA technology such as quantification of DNA, isolation of chromosomal DNA, isolation of plasmid DNA from bacterial cells, restriction digestion of DNA and their separation using Agarose gel electrophoresis, amplification of DNA fragment by PCR. With learning these techniques students will gain expertise to work further in the area of recombinant DNA technology.
THEORY
UNIT I (10 Periods)
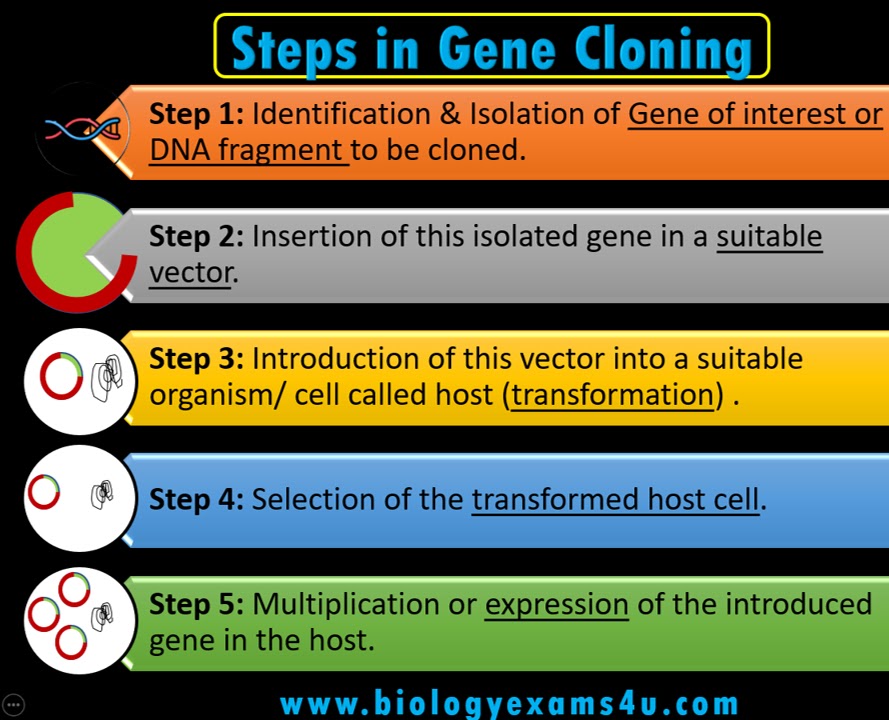
Molecular tools and applications- restriction enzymes, ligases, polymerases, alkaline phosphatase. Gene Recombination and Gene transfer: Transformation, Episomes, Plasmids and other cloning vectors (Bacteriophage-derived vectors, artificial chromosomes), Microinjection, Electroporation, Ultrasonication, Principle and applications of Polymerase chain reaction (PCR), primer-design, and RT- (Reverse transcription) PCR.
UNIT II (10 Periods)
Restriction and modification system, restriction mapping. Southern and Northern hybridization. Preparation and comparison of Genomic and cDNA library, screening of recombinants, reverse transcription, Genome mapping, DNA fingerprinting, Applications of Genetic Engineering Genetic engineering in animals: Production and applications of transgenic mice, role of ES cells in gene targeting in mice, Therapeutic products produced by genetic engineering-blood proteins, human hormones, immune modulators and vaccines (one example each).
UNIT III (10 Periods)
Random and site-directed mutagenesis: Primer extension and PCR based methods of site directed mutagenesis, Random mutagenesis, Gene shuffling, production of chimeric proteins, Protein engineering concepts and examples (any two).
UNIT IV (10 Periods)
Genetic engineering in plants: Use of Agrobacterium tumefaciens and A. rhizogenes, Ti plasmids, Strategies for gene transfer to plant cells, Direct DNA transfer to plants, Gene targeting in plants, Use of plant viruses as episomal expression vectors.
PRACTICALS
1. Isolation of chromosomal DNA from plant cells
2. Isolation of chromosomal DNA from E.coli
3. Qualitative and quantitative analysis of DNA using spectrophotometer
4. Plasmid DNA isolation
5. Restriction digestion of DNA
6. Making competent cells
7. Transformation of competent cells.
8. Demonstration of PCR
SUGGESTED READING
1. Brown TA. (2006). Gene Cloning and DNA Analysis. 5th edition. Blackwell Publishing, Oxford, U.K.
2. Clark DP and Pazdernik NJ. (2009). Biotechnology-Applying the Genetic Revolution. Elsevier Academic Press, USA.
3. Glick, B.R., Pasternak, J.J. (2003). Molecular Biotechnology- Principles and Applications of recombinant DNA. ASM Press, Washington
4. Primrose SB and Twyman RM. (2006). Principles of Gene Manipulation and Genomics, 7th edition. Blackwell Publishing, Oxford, U.K.
5. Sambrook J, Fritsch EF and Maniatis T. (2001). Molecular Cloning-A Laboratory Manual. 3rd edition. Cold Spring Harbor Laboratory Press.
- Teacher: Annika Singh